





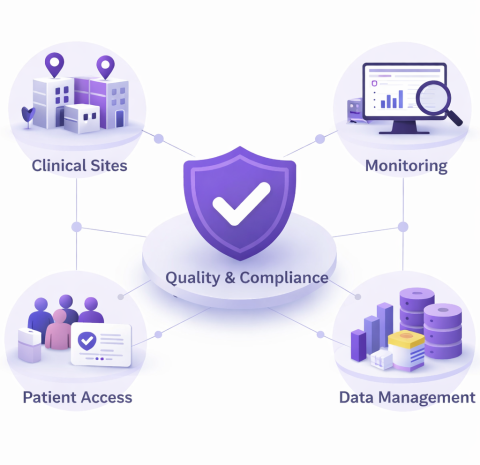
Why Choose MCT?
Extensive Patient Access Across the Region
Broad Therapeutic Experience Across Study Phases
Established Partnerships with Key Clinical Sites
Proven Record of Audit Compliance & Quality Excellence

ALAN OEAN, CO-FOUNDER

About us
Different types of high-quality
services department
- Pathology Testing
- Chemical Research
- Healthcare Labs
- Sample preparation
- Environmental Testing
- Advances Microscopy

Regulatory Compliance
MCT ensures full regulatory compliance across all clinical studies conducted in the Middle East, Africa, Pakistan, and the Eurasian Economic Union (EAEU).
Our operations are governed by robust Standard Operating Procedures (SOPs) and fully aligned with international Good Clinical Practice (GCP) guidelines. By combining global standards with strong local expertise, we help clients navigate complex and evolving regulatory environments—from initial submissions through site activation and close-out, including during regulatory inspections.
Key Strengths:
- Compliance: Adherence to international GCP and regional regulatory guidelines
- SOPs: Robust Standard Operating Procedures applied across all studies in line with international standards
- Regulatory Intelligence: Staying up to date with global regulatory developments and assessing their local impact
- Inspection Readiness: Managing client engagement with regulatory authorities and ensuring inspection readiness
- Proven Track Record: 100% of MCT studies completed with no critical regulatory findings.

Regulatory Compliance
MCT ensures full regulatory compliance across all clinical studies conducted in the Middle East, Africa, Pakistan, and the Eurasian Economic Union (EAEU).
Our operations are governed by robust Standard Operating Procedures (SOPs) and fully aligned with international Good Clinical Practice (GCP) guidelines. By combining global standards with strong local expertise, we help clients navigate complex and evolving regulatory environments—from initial submissions through site activation and close-out, including during regulatory inspections.
Key Strengths:
- Compliance: Adherence to international GCP and regional regulatory guidelines
- SOPs: Robust Standard Operating Procedures applied across all studies in line with international standards
- Regulatory Intelligence: Staying up to date with global regulatory developments and assessing their local impact
- Inspection Readiness: Managing client engagement with regulatory authorities and ensuring inspection readiness
- Proven Track Record: 100% of MCT studies completed with no critical regulatory findings.

Regulatory Compliance
MCT ensures full regulatory compliance across all clinical studies conducted in the Middle East, Africa, Pakistan, and the Eurasian Economic Union (EAEU).
Our operations are governed by robust Standard Operating Procedures (SOPs) and fully aligned with international Good Clinical Practice (GCP) guidelines. By combining global standards with strong local expertise, we help clients navigate complex and evolving regulatory environments—from initial submissions through site activation and close-out, including during regulatory inspections.
Key Strengths:
- Compliance: Adherence to international GCP and regional regulatory guidelines
- SOPs: Robust Standard Operating Procedures applied across all studies in line with international standards
- Regulatory Intelligence: Staying up to date with global regulatory developments and assessing their local impact
- Inspection Readiness: Managing client engagement with regulatory authorities and ensuring inspection readiness
- Proven Track Record: 100% of MCT studies completed with no critical regulatory findings.

Regulatory Compliance
MCT ensures full regulatory compliance across all clinical studies conducted in the Middle East, Africa, Pakistan, and the Eurasian Economic Union (EAEU).
Our operations are governed by robust Standard Operating Procedures (SOPs) and fully aligned with international Good Clinical Practice (GCP) guidelines. By combining global standards with strong local expertise, we help clients navigate complex and evolving regulatory environments—from initial submissions through site activation and close-out, including during regulatory inspections.
Key Strengths:
- Compliance: Adherence to international GCP and regional regulatory guidelines
- SOPs: Robust Standard Operating Procedures applied across all studies in line with international standards
- Regulatory Intelligence: Staying up to date with global regulatory developments and assessing their local impact
- Inspection Readiness: Managing client engagement with regulatory authorities and ensuring inspection readiness
- Proven Track Record: 100% of MCT studies completed with no critical regulatory findings.

Regulatory Compliance
MCT ensures full regulatory compliance across all clinical studies conducted in the Middle East, Africa, Pakistan, and the Eurasian Economic Union (EAEU).
Our operations are governed by robust Standard Operating Procedures (SOPs) and fully aligned with international Good Clinical Practice (GCP) guidelines. By combining global standards with strong local expertise, we help clients navigate complex and evolving regulatory environments—from initial submissions through site activation and close-out, including during regulatory inspections.
Key Strengths:
- Compliance: Adherence to international GCP and regional regulatory guidelines
- SOPs: Robust Standard Operating Procedures applied across all studies in line with international standards
- Regulatory Intelligence: Staying up to date with global regulatory developments and assessing their local impact
- Inspection Readiness: Managing client engagement with regulatory authorities and ensuring inspection readiness
- Proven Track Record: 100% of MCT studies completed with no critical regulatory findings.

Regulatory Compliance
MCT ensures full regulatory compliance across all clinical studies conducted in the Middle East, Africa, Pakistan, and the Eurasian Economic Union (EAEU).
Our operations are governed by robust Standard Operating Procedures (SOPs) and fully aligned with international Good Clinical Practice (GCP) guidelines. By combining global standards with strong local expertise, we help clients navigate complex and evolving regulatory environments—from initial submissions through site activation and close-out, including during regulatory inspections.
Key Strengths:
- Compliance: Adherence to international GCP and regional regulatory guidelines
- SOPs: Robust Standard Operating Procedures applied across all studies in line with international standards
- Regulatory Intelligence: Staying up to date with global regulatory developments and assessing their local impact
- Inspection Readiness: Managing client engagement with regulatory authorities and ensuring inspection readiness
- Proven Track Record: 100% of MCT studies completed with no critical regulatory findings.
Our Services
Delivering clinical research solutions across the Middle East, Africa, Pakistan and EAEU





50 +
Countries Covered
570 +
Sites
1950 +
Investigators
27000 +
Patients Enrolled
Testimonials
What our clients and collaborators say about their experience working with MCT
I have been working with MCT for the past two years now on clinical research projects, and I can confidently say their support has been exceptional. From regulatory submissions to data management and site coordination, their team has consistently shown professionalism, reliability, and expertise. They are always easy to reach and their attention to detail have allowed our research to run smoothly and on schedule.
Professor of Clinical Oncology
Gustav Roussy International Egypt, CairoWorking with MCT has always given us the comfort and safety we eagerly need as investigator and study team. Their meticulous management and superb attention to detail evoke trust and positions them as the right partner to ensure clinical trials are conducted to the highest quality standards, especially that study sponsors and patients expect nothing less.
Chief Research Officer
Burjeel Medical City, UAE- Expertise.
- Research.
- Compliance.
- Quality
- Integrity.
- Collaboration.
- Excellence.
- Diversity.
- Therapeutics.
Why Conduct Clinical Trials in the MIDDLE EAST, AFRICA, Pakistan & EAEU?
These regions represent one of the most underutilized yet high-potential environments for clinical research, offering access to large, diverse, and often treatment-naïve patient populations.
High recruitment potential and genetic diversity
Leading healthcare institutions and experienced investigators
Strong disease relevance and growing investment in research
Laboratories used for scientific research take many forms because of the differing requirements of specialists in the various fields of science and engineering with laboratory particle accelerator
Laboratories used for scientific research take many forms because of the differing requirements of specialists in the various fields of science and engineering with laboratory particle accelerator
Laboratories used for scientific research take many forms because of the differing requirements of specialists in the various fields of science and engineering with laboratory particle accelerator
Best Outcome For Every Patient Every Time or contact us via support@labtechco.com
Ready to advance your clinical trials with local regional expertise?
Latest News & Blogs
atem accusantium doloremque laudantium.



